-
Life Sciences
.png)
Our solutions
-
Materials Analysis
.png)
Our solutions
Techniques
Applications
- Why Delmic?
-
Insights
.png)
Insights
-
Company
.png)
Company
Life Sciences
How New York Structural Biology Center increased their lamellae production 3x without compromising the workflow

Automation and high throughput in single particle analysis and tomography are the goals of many structural biology research centers, especially for a global leader like the New York Structural Biology Center. Armed with the CERES Ice Shield, the New York Center was able to protect lamellae from ice contamination and achieve both an increase in lamellae production and resolution.
BACKGROUND
The customer
New York Structural Biology Center (NYSBC) is a world leader in structural biology. Housed within NYSBC, the National Center for In-situ Tomographic Ultramicroscopy (NCITU) operates as both a service center for sample preparation and production of thin specimens suitable for cryogenic electron tomography (cryo-ET) and a provider for access and training users in the development of the skills needed in cryo-ET. One overarching goal at NYSBC is automation and high throughput both in single particle analysis and tomography. To that end, the service center works to maximize lamellae production.
The techniques and equipment
A standard in-situ workflow at NCITU starts with sample preparation and ends with high-resolution cryo-ET data collection. The center provides facilities for cell growth, target protein expression, and live cell screening. To preserve the state of the biological system, cells are vitrified by either plunge-freezing or high-pressure freezing. The center developed and advanced a lamella preparation technique that works with the latter method, known as the ‘waffle’ method. According to Reza Paraan (Postdoctoral Researcher, NYSBC), this method generates consistently longer lamella, which helps NCITU reach its goal of getting high numbers of tomograms for higher resolution reconstruction of protein complexes.
With a focus on cells grown on grids as well as in high-pressure frozen samples, NCITU receives proposals from all over the United States for access and training on Aquilos 2, a cryogenic focused ion beam/scanning electron microscope (FIB/SEM) that is dedicated to sample preparation. The microscope is cooled down using a SubAngstrom's dewar solution. Reza has access to the system at NCITU to carry out his research.
BOTTLENECK
The main bottleneck for high-throughput lamellae production was the recontamination rate. Initially, it was 20 nm/hr. The build-up of amorphous ice during an overnight milling session would cause the lamellae to increase in thickness and the outcome was undesirable.
At first, NCITU got a fix from their supplier on the pole piece that reduced the contamination to 5 nm/hr. However, the ice build-up overnight was still significant. Another alternative was tried. “We switched to shorter milling sessions and that reduced the throughput”, Reza Paraan recalls.
We were limited to milling during the day with a tight schedule, and no overnight milling.
SOLUTION
To effectively prevent amorphous ice growth, NCITU knew that they needed something more effective without changing their workflow. After careful consideration and a thorough feasibility study, Delmic's CERES Ice Shield was chosen due to its compatibility with cryo-FIB/SEM and the 'waffle' method's workflow, making it easy for the center to implement the shutter in the current system.
Delmic’s ice contamination minimization solution, CERES Ice Shield, is liquid nitrogen-cooled and provides cryo-pumping to lower the partial pressure of water in the sample vicinity, thus reducing ice build-up to a non-measurable level. After installation, the CERES Ice Shield reduced the contamination rate to 1.5 nm/hr or less.
Another feature that has proven to be a great benefit is its running time. CERES Ice Shield runs as long as the cryo stage’s duration. Combined with the subangstrom dewar’s power, according to Reza, it prolongs the microscope operation time to two to three days with 24 hours of milling.
RESULTS
With almost no contamination build-up, CERES Ice Shield enables Reza and other users of NCITU to mill many more lamellae in one session and free them from tight milling schedules. That gives them more time to experiment with more difficult samples and increase the achievable resolution.
Moreover, the increase in throughput is significant:
- Cells grown on grids: 2-fold increase
- Waffled samples: 3-fold increase
This means not only an increase in the number of lamellae that NCITU produces but also an increase in the percentage of high-quality lamellae. “As an example, we were able to generate 12 lamellae on a grid with high-pressure frozen ribosomes (using the waffle method for purified proteins),” Reza says. “This enables us to collect a single particle data on that grid. This wouldn’t have been possible with a smaller number of lamellae.”
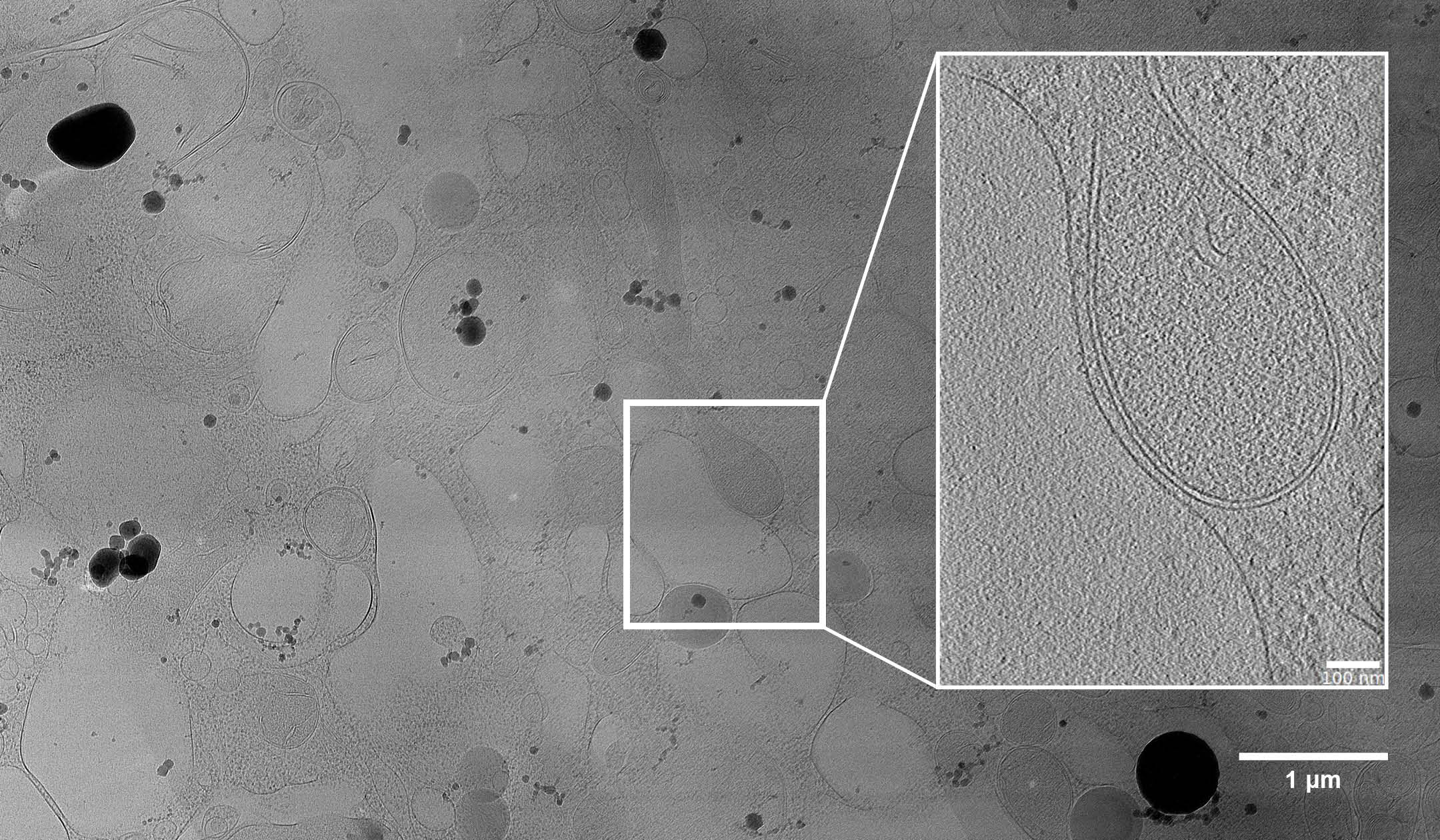
Figure 1: A lamella of the ER-mitochondria contact site, prepared using CERES Ice Shield. High contrast can be seen due to the reduced ice contamination.
Generally, having more time with the sample in the chamber results in higher throughput or more flexibility, which is one of the overarching goals at NYSBC. NYSBC’s case proves that CERES Ice Shield, implemented in NCITU, has allowed them to make breakthroughs in structural biology that they were not able to before. Most importantly, Reza and NYSBC’s trust in Delmic reflects that we deliver on what we promise:
The nicest part is that their products live up to their specifications and are offered at a competitive cost.




