-
-
CL Solutions
Cathodoluminescence solutions that reveal fundamental properties of matter
-
Cryo Solutions
Integrated and automated solutions to u nlock the power of cryo - ET workflow -
FAST IMAGING
Fast EM solutions for reliable and high throughput electron microscopy
-
CLEM Solutions
Integrated correlative microscopy solutions that combine the power of fluorescence and electron microscopy
-
CL Solutions
- Contact
- All resources
-
CL Solutions
Cathodoluminescence solutions that reveal fundamental properties of matter
-
Cryo Solutions
Integrated and automated solutions to u nlock the power of cryo - ET workflow -
FAST IMAGING
Fast EM solutions for reliable and high throughput electron microscopy
-
CLEM Solutions
Integrated correlative microscopy solutions that combine the power of fluorescence and electron microscopy
Fast em solutions
High-throughput electron microscopy: frequently asked questions
Answering the most common questions about high-throughput electron microscopy and our ultra-fast multibeam SEM solutions.
High-throughput electron microscopy
-
How does Delmic’s high-throughput scanning electron microscope (SEM) FAST-EM enable fast imaging?
FAST-EM’s high throughput is achieved by a combination of several technological features. The system is a multibeam SEM, capable of imaging with 64 beams in parallel. Combined with highly automated software and robust microscope components, the system sustains throughputs of up to a hundred times higher than a conventional electron microscope. See below some unique features of the system:
- High resolution, high-contrast images acquired with transmission electron detection
- Easy acquisition procedure with continuous operation for a minimum of 3 days
- Sustained throughput of 100 megapixels per second during routine imaging at 400 ns dwell time
Learn more about FAST-EM
-
What are the benefits of collecting both contextual and high-resolution data in electron microscopy?
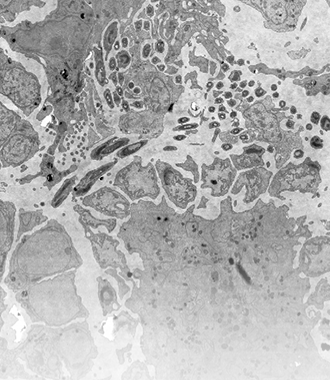
A high-speed electron microscope combined with an automated workflow can eliminate the bottleneck created by the throughput of conventional EMs. Sections can be imaged in their entirety, after which the full, high-resolution dataset is available for (automated) analysis. In addition, this way data is acquired in an unbiased manner. Datasets contain the full context of the cell or tissue, rather than a context provided by a (potentially) biased researcher.
Learn more in the blog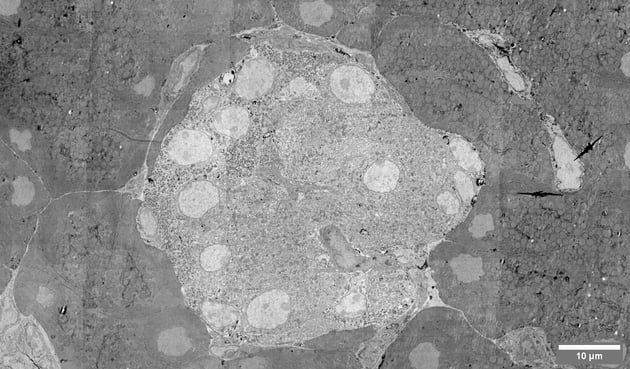
-
How does FAST-EM benefit volume electron microscopy?
Volume EM can benefit directly from the high-speed electron microscopes, as larger volumes can be imaged within a smaller amount of time. At the same time, high-throughput imaging enables comparative studies, which were previously too time-consuming to be feasible.
Learn more about volume EM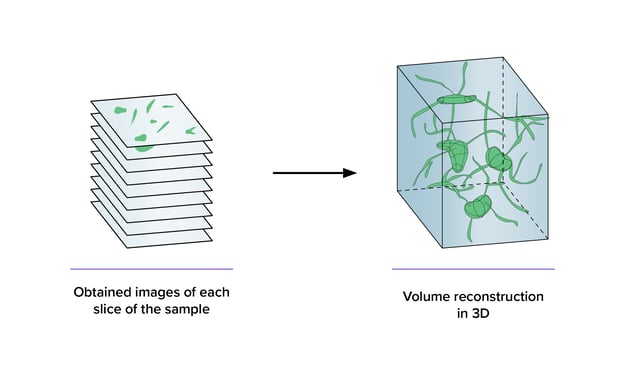
-
How can large scale electron imaging projects benefit from high-throughput microscopy?
Faster imaging enables a rapid, unbiased method of data collection, which is crucial for large-scale imaging projects. Moreover, performing fast imaging on large samples, such as whole tissues and organs, can gain both nanoscale information needed to analyze subcellular details and the context needed to understand the distribution of cell types within tissues or organs.
Learn more about high throughput EM for large scale imaging projects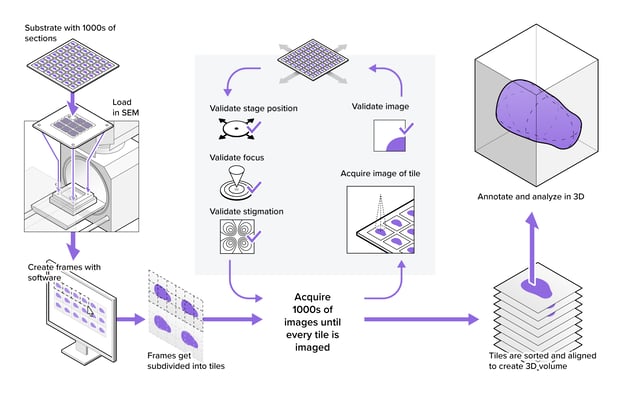
Applications
-
Which research fields can benefit from high-throughput electron microscopy?
High-throughput EM is extremely beneficial for many research fields, including neurobiology, cell biology, histology, plant biology, digital pathology, analysis of tissues, cells.
-
How can high-throughput EM unlock the potential of CLEM?
In traditional CLEM workflows, EM is a limiting factor for the scope of data collected. With FAST-EM imaging the samples 100 times faster, CLEM workflows can be enormously simplified and researchers can observe fine morphological details while retaining the overview that is normally only collected with fluorescence microscopy. In addition, larger volumes can be investigated by CLEM, enabling a transformation towards integration of imaging modalities on a large scale.
Learn more about advancing CLEM with high-throughput EM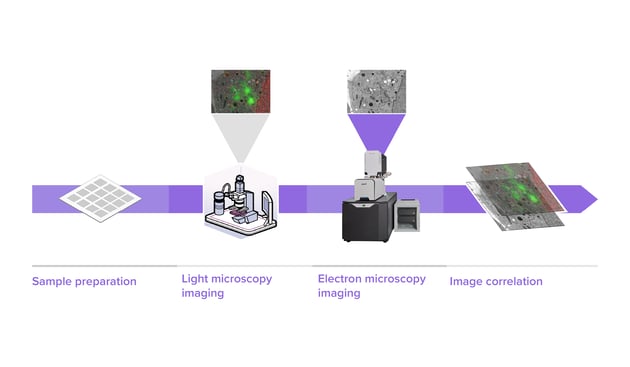
-
What are the benefits of using high-throughput EM in cancer research?
All the six main hallmarks in the development of human tumours can be observed by using 3D high-resolution imaging. Other than that, the high resolution of 3D electron microscopy (3DEM) is also highly useful for detecting minimal changes in cells and tissues as a diagnostic technique in cancer research. Together with the use of microscopic X-ray computed tomography, the 3DEM images of the tumour cells can be used to study the ultrastructure of these cells in vivo.
Learn more about this application
-
What are the benefits of using high-throughput EM in connectomics?
With high-throughput EM is it possible to collect volumes of interest much faster, which enables neuronal tracing over large distances and a better understanding of cellular interactions in the brain and other tissues. The decrease in time to acquire large 3D images enables imaging of multiple specimens within a single project. This opens the possibility to compare the same specimen under different experimental conditions, like genetic modification, or drug or ablation treatments. Multiple aspects of Delmic’s Fast Imaging workflow are beneficial for the connectomics field, like its straightforward sample preparation, easier data analysis and the highly automated acquisition pipeline.
Learn more about this application
-
How can high-throughput EM help my work in an imaging facility?
Samples can be imaged automatically at high speed, while region of interest (ROI) selection and data analysis can be performed offline (away from the microscope). Since a microscope operator is no longer needed to identify relevant biological structures at the microscope, many or larger samples can be imaged within the same amount of time. Moreover, FAST-EM can run unattended for 3 days, significantly increasing productivity and cost-efficiency.
Learn more about the benefits
Sample preparation
-
What’s the basic protocol of sample preparation for high-throughput EM?
Samples for high-throughput EM can be prepared based on the routine for biological electron microscopy projects, therefore no, or only minimal, alterations are needed. A basic protocol involves at least the following steps:
1. Chemical fixation of the sample with fixatives such as glutaraldehyde and paraformaldehyde.
2. Staining with heavy metals such as osmium, lead, and uranium
3. Dehydration through a graded ethanol or acetone series
4. Resin embedding of the sample.
5. Ultramicrotome sectioning to a suitable thickness (<150 nm)
6. Placement of sections on substrates. -
What are the staining protocols for high-throughput EM workflows?
Osmium, uranyl and lead stainings have been the standards of electron microscopy experiments for the last few decades. To match new applications and EM systems, there are also adaptations of new protocols, such as enhanced staining protocols and mild staining protocols.
Read more about the staining protocols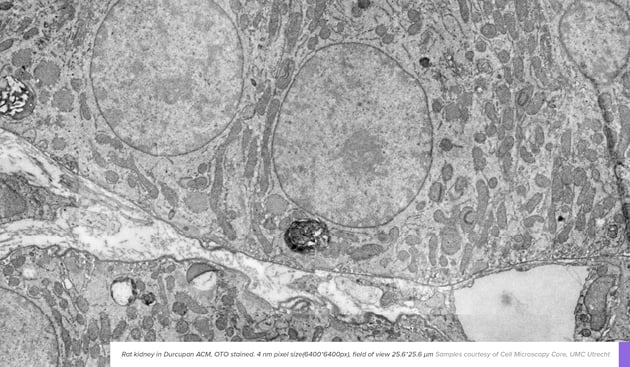
-
How can I handle samples for FAST-EM?
FAST-EM uses custom scintillator substrates as sample support, which are an integral part of the system. Like other EM substrates such as silicon wafers or ITO-coated glass slides, sections can be loaded onto the scintillators in a variety of ways. Individual sections are easily collected with a pickup loop. Ribbons of multiple sections are collected by substrate in the water bath of the microtome knife and then draining the water out, leaving the sections in place on the substrate. Altogether, only minimal changes are required to your workflow to produce samples for FAST-EM.


Didn't find an answer?
Fill in the form with your question, and our experts will get back to you soon!
